|
Nonmetals are elements that cannot conduct heat or electricity.Metals easily lose electrons and form positive ions (cations). Metals are elements that effectively conduct heat and electricity.Metallic character is defined by the degree of reactivity of a metal.Electron affinity is a measure of a neutral atom’s strength of gaining an electron.Electronegativity is a measure of the strength of atoms in attracting a shared pair of electrons to themselves. 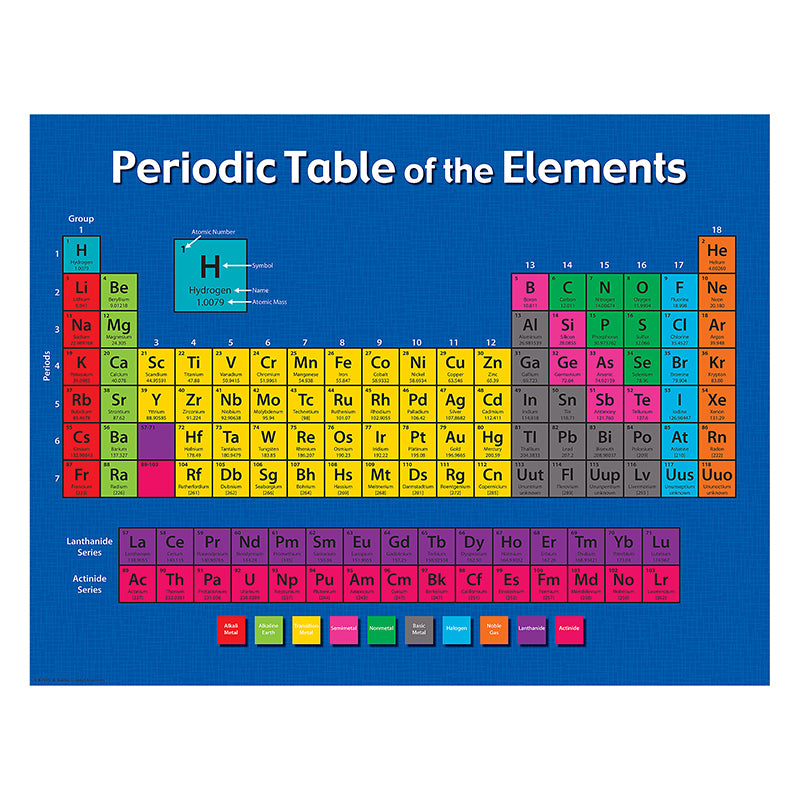
Ionization energy is the energy needed to remove electrons from a neutral atom resulting in a positively charged ion.Electron configuration is the distribution of electrons in atomic or molecular orbitals.The elements are also grouped into metals, metalloids, and nonmetals.The blocks are labeled the s-block, f-block, d-block, and p-block.Groups are numbered from 1 (leftmost column) to 18 (rightmost column).  
There are seven rows (periods), 18 columns (groups), and four blocks in the periodic table.The organization of elements can also predict the properties of unknown or newly discovered elements.The arrangement of chemical elements can provide insight about the relationships of the elements with each other.Atomic number – the number of protons in the atom’s nucleus.Name – the common name of the element.Symbol – an abbreviation of the chemical element’s name.The parts included in the element’s label depends on the version of the periodic table, but these are the basics:.The periodic table we use now is based on Mendeleev’s structure.Deming was being circulated in American schools. By the 1930s, the popular version of the periodic table attributed to Horace G.In 1871, Russian Chemist Dmitri Mendeleev published a new edition of his periodic table with similar elements grouped in columns rather than in rows.From 1863 to 1866, English chemist John Newlands wrote a series of papers explaining similar properties of elements recurring at intervals of eight, which became known as the Law of Octaves.In 1864, German chemist Julius Lothar Meyer published a table with 28 elements while English chemist William Odling published a table of 57 elements.In 1862, French geologist Alexandre-Emile Beguyer de Chancourtois published the “telluric screw” which is a three-dimensional arrangement of the elements.In 1857, French chemist Jean-Baptiste Dumas described relationships between groups of metals in his published work.In 1843, German chemist Leopold Gmelin produced a table of 55 elements which is one of the foundations of our modern periodic table.In 1829, German chemist Johann Wolfgang Döbereiner discovered that the elements can be grouped into triads based on their chemical properties, which became known as the Law of Triads.In the following century, chemists continued to research and observe relationships of the elements and develop one scheme that unites these relationships. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
